Friday 10/April/2026 – 06:04 AM
Investigations into the case of accusing 10 officials at the 6th of October Health Insurance Hospital in Dokki of causing 75 patients to lose their sight revealed new details through the statements of one of the defendants in the case.
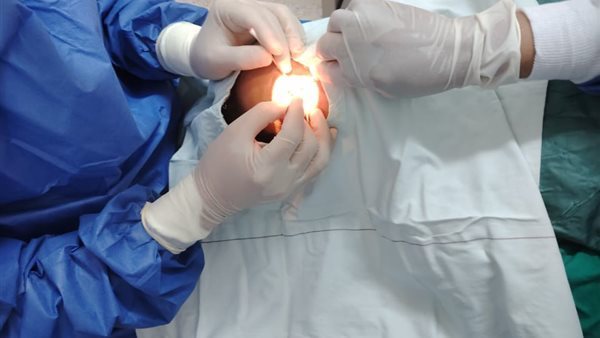
An incident of 75 patients becoming blind inside the 6th of October Health Insurance Hospital
Cairo 24 obtained details of M.’s confessions. 41 years old, former director of the 6th of October Health Insurance Hospital in Dokki. When asked about whether complaints were issued regarding infection control throughout your tenure as director of the 6th of October Hospital in the Ophthalmology Department? He replied: No.
The rest of the details of his statements regarding the incident were as follows:
Q: Is there a limit on the number of surgeries per day in the hospital’s ophthalmology department?
A: On average, there are 30 cases per day between cataract surgery and injections.
Q: Is there a maximum limit for surgeries from the Health Insurance Authority?
A: No, but we were last at 6 pm.
Q: What is the time period between each surgery and the other for ophthalmology surgeries in the hospital?
A: It is approximately 10 minutes or a quarter of an hour, and he specifically asks the hospital’s quality management about that.
Q: How many rooms are allocated for ophthalmology surgeries in the hospital?
A: Two operating rooms, one in which cataract, glaucoma, and corneal operations are performed, and the second room is for injections only.
Q: Is anesthesia given in the same surgery room?
A: No, the anesthesia is local in the recovery room inside the operating room.
Q: What are the procedures followed in the event that abnormal infection rates are recorded in the hospital?
A: The followed procedure is that the infection control committee notifies me according to the procedure, and I take swabs from the department where the infection is located, take isolation measures if imposed, prepare the necessary treatment protocol and review sterilization procedures, and if it is proven that there is an infection, a report is prepared and submitted to my presidency.
Q: Are samples taken periodically to be tested to determine the presence of infection or not?
A: Throughout my time as director, no swab test has been performed, as far as I know, and this is due to the infection control committees.
Q: Were you informed of a bacterial infection in any department of the hospital during your tenure?
A: As of the date of the incident, I have not been informed of anything.
Q: When did the infection begin to spread specifically among patients?
A: The infection began to appear on 8/31/2025, and the ophthalmology doctor (M.A.) suspected it was methylfoam. However, on 9/1/2025, more cases appeared in the clinic with the same symptoms, and accordingly, as hospital director, I stopped ophthalmology operations and assigned the infection control officer to take all precautions and investigate the causes of this infection.
Q: What is the surgical intervention that caused the infection under investigation?
A: All of the cases that came with these infections were cases of removing cataracts from the eyes, which confirmed to us that the problem was due to these operations.
Q: How exactly is cataract removal performed and what medical devices are used for it?
A: I do not know the procedure procedures, and the specialist doctor will ask about that. The device used is the phaco device.
Q: What is the suitability of the “Phaco” device for working on such surgical interventions?
A: It is any device that has a lifespan, but the idea of whether the device is used or not is up to the medical engineer.
Q: Since when has the “Phaco” device been used at the 6th of October Hospital for health insurance?
A: This phaco device was supplied in 2013 and was used in 2014.
Q: Are there regular maintenance performed on that device?
A: There are 3 Phaco devices in the hospital, one of which is a near-prosthesis device that has not been used, and two other devices that are used interchangeably. However, on June 1, 2025, a malfunction occurred in one of the two devices and it was withdrawn from the hospital by the maintenance company contracting with health insurance. The other device was tested and confirmed to be fit to work. The chief nurse and responsible doctors were trained to work with it and the doctor (M.A.) signed off on the validity of the device.
Q: Did the company responsible for maintenance make any recommendations after reviewing the device?
A: I have not been informed of any recommendations from the company.
Q: When did the aforementioned device start working?
A: The first device was damaged on 6/1/2025, and the other device began working from 6/3/2025 until the date of stopping operations on 9/1/2025.